
ASHP members are contributing pharmacists’s point of view to a number of national healthcare quality efforts.
MEASURING, MONITORING, AND IMPROVING PATIENT CARE is becoming increasingly important in today’s healthcare environment, and the opportunity for pharmacists to influence the quality measures that are used has never been greater.
Multi-stakeholder groups such as the National Quality Forum (NQF) are a vital part of this process, endorsing standards for performance measurement and validating quality measurements used in federal payment programs. Through ASHP’s Quality Advocates, pharmacist participation in these groups is helping to shape the reimbursement landscape and improve patient care.
“Our members help build consensus in the rigorous environment of the steering committees,” said Shekhar Mehta, Pharm.D., M.S., ASHP’s director of clinical guidelines and quality improvement. “They assess the feasibility, reliability, validity, and scientific acceptability of proposed measures, and other committee members value pharmacists’ ideas on how a given measure could be implemented in real practice.”
Immediate Impact
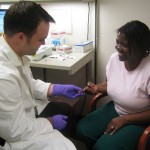
It didn’t take long for Starlin Haydon-Greatting, M.S., B.S.Pharm., FAPhA, clinical pharmacist consultant at SHG Clinical Consulting and the IPhA’s Patient Self-Management Program in Springfield, Ill., to have an impact on NQF’s endorsement process. She began her two-year term on the Endocrine Steering Committee in January, and she has already provided influential feedback on several measures that address drug adherence. Haydon-Greatting partnered with another committee member, a researcher for a pharmaceutical company, to explain ways of calculating adherence to other committee members.

Starlin Haydon-Greatting, M.S., B.S.Pharm., FAPhA, is working on drug adherence measures as part of the NQF’s Endocrine Steering Committee.
“Clinicians such as nurse practitioners want to know how they can ensure that patients are adhering to the prescriptions they write. They generally don’t know if the patient has been taking medications until the next visit, so they want to know how to create [patient] accountability,” Haydon-Greatting said.
“I made the point that pharmacists can see if patients are filling their prescriptions, and that pharmacists can take the lead on tracking that kind of data.”
Haydon-Greatting drew upon her work as pharmacy network coordinator for Taking Control of Your Health, an offshoot of the Diabetes 10-City Challenge in which pharmacists lead treatment programs for employees with diabetes.
“We have electronic medical records and web-based programs, and I was able to speak from experience and say that once you have those, the data is already there.”
She added that her input has not only been well-received, but actively solicited by other members of the group. “There are over 20 physicians on this committee, and once they found out I was the only pharmacist, every time a question came up about medication use or adherence, all heads turned to me.”
Winning Recognition
Haydon-Greatting’s experience may be testimony to how far pharmacists have come in the eyes of other clinicians. Five years ago, Steven M. Riddle, Pharm.D., BCPS, FASHP, director of clinical development for Pharmacy OneSource/Wolters Kluwer Health in Seattle, had to prove his mettle on the NQF’s Ambulatory Care Steering Committee.
“I was on a panel with some well-known people in positions of authority [in their fields], national leaders who were not easy-going, subtle folks. There I was, [then] just a pharmacist from the University of Washington to them, and they challenged me. I had to earn a little credibility,” Riddle said.
He earned their respect by applying his background and training as a pharmacist to the four key criteria the committee used in deciding whether to endorse a measure—its importance, scientific acceptability, usability, and feasibility.
“Pharmacists are trained in evidence-based medicine, understanding the trials, and determining whether the clinical and technical evidence is valid. That’s part of what [NQF Committees] must look at when evaluating a measure,” said Riddle, who served until recently the chair for ASHP’s Section of Ambulatory Care Practitioners. “There were times when I swayed opinion by going back to the four criteria, but it was tricky to negotiate. For example, something could be highly valuable but completely unfeasible.”
As with Haydon-Greatting, once Riddle demonstrated his knowledge, the other committee members were quick to tap him for input. “I was able to hold my own and bring forward my concerns. Then when they had questions about medication use, they would ask me, ‘Well, what do you think, Steve?’ ”
Looking to the Future
Pharmacists have their work cut out for them on these committees, said Jannet Carmichael, Pharm.D., FCCP, FAPhA, BCPS, VISN 21 Pharmacy Executive at the Department of Veterans Affairs in Reno, Nev. She encourages young pharmacists to look into systems development and healthcare analytics, noting how her experience with the VA’s clinical data warehouse proved invaluable when she served on the NQF’s Medication Management Steering Committee in 2009.
“The VA is a data utopia. Knowing that I came from an environment with a mature electronic medical record system, the committee members were willing to give me a bit of a bye in presenting my views on evaluating and collecting health data,” she said.
“As electronic medical records become the norm, and the business of metrics and quality measurements becomes more important, the ability to load data sets and analyze the information will become essential.”
Recognizing the need for pharmacist representation in groups like the NQF, ASHP is bolstering efforts for more participation among its membership.
“We’ve been trying to bring more pharmacists into the various committees,” said Christopher J. Topoleski, ASHP’s director of federal regulatory affairs. “Younger pharmacists are more and more interested in informatics. They’re a tech-savvy generation, and as they get experience in using data in the implementation of quality improvement measures, we’ll have a larger crop of people to choose from.”
–By Terri D’Arrigo
Editor’s note: The above story is the second part of a two-part series on how ASHP members are influencing and steering national quality measures. Click here to read the first story.








 If you want to contribute tutorials, news or other stuff please contact us. We pay 150 for each approved article.
If you want to contribute tutorials, news or other stuff please contact us. We pay 150 for each approved article. Consectetur adipisicing elit. Sed do eiusmod tempor incididunt ut labore.
Consectetur adipisicing elit. Sed do eiusmod tempor incididunt ut labore. This site uses valid HTML and CSS. All content Copyright © 2010 Newscast, Inc
This site uses valid HTML and CSS. All content Copyright © 2010 Newscast, Inc If you like what we do, please don't hestitate and subscribe to our
If you like what we do, please don't hestitate and subscribe to our